
Suppose you’re a new researcher who hasn’t yet been exposed to the vast array of regulations, reports, and other requirements imposed on federal grants. You wonder what you might be facing if you manage to get an NIH grant.
So you start by looking at the 103 pages of rules on how to spend and account for federal funds, how to be prepared for audits, and the like. You quickly move on, because your university has a grants/accounting department that will handle most or all of this.
But that was just the starting point. Now you turn to the “Grants Policy Statement” for NIH, only to find that it is literally 420 pages long.
Indeed, the first 44 pages just define all of the terms and abbreviations used in the rest of the document.
This doesn’t bode well if you were hoping for a quick evening.
In this seemingly endless document, you’ll find all sorts of rules and expectations, with a very tiny fraction including things like the following:
- If an undergraduate spends one month or more working on your project, you will have to make them register in the NIH’s online system and submit complete information, even if they aren’t actually paid from the NIH grant (section 2.2.1.3).
- If you fly to an international conference, you have to use a “U.S. flag air carrier” that “holds a certificate under 49 U.S.C. § 41102” (I’m sure the average biology professor is totally up to date on what that means). Helpfully, you can get around this requirement if it would require a layover of “four hours or more at an overseas interchange point” or if it would “double the travel time” on a trip of three hours or less, so plan to spend even more time documenting all of that (section 4.1.11).
- Executive Order 12770 (July 25, 1991) requires that you use the metric system in any applications, reports, and publications (section 4.1.18). No miles or gallons in your application.
- Under the Pro-Children Act of 1994 (P.L. 103-227), you can’t allow smoking in any building used for school or childcare, except for facilities funded solely by Medicaid, or used for inpatient drug/alcohol treatment, or facilities where WIC coupons are redeemed (section 4.1.23).
- Under Executive Order No. 13043 (April 16, 1997), you are “encouraged” to adopt seat belt “policies and programs” for anyone who is driving a vehicle while on the job (section 4.1.28).
- Under Executive Order 13513, you should adopt rules and programs to “prohibit text messaging while driving” (section 4.1.31).
- You cannot “advocate or promote gun control” (section 4.2.4).
- If you move to a different university (professors are known to do this from time to time), you have to get NIH’s prior approval, and submit an “Official Statement Relinquishing Interests and Rights in a Public Health Service Research Grant” from the first university, and then make sure that the second university submits around 20 forms (section 8.1.2.7).
- You should submit an “Individual Development Plan” for any graduate student or post-doc supported by your grant (Section 8.4.1.1.2).
As I say, these are but a tiny fraction of the overall set of rules and requirements. There are many more.
But here’s the thing: All of these requirements are well-motivated.
If you think about each of these requirements in isolation, then of course it makes sense that we wouldn’t want people to text while driving or fail to wear a seat belt, now would we? And we wouldn’t want graduate students and post-docs to be treated as menial employees with no future development in sight. And we wouldn’t want to let grantees switch universities without making sure that the old and the new university are both going to play ball. And we wouldn’t want to hand out federal dollars without requiring all sorts of reporting and controls, now would we?
And so on, ad infinitum.
But put all these requirements together, and it is death by a thousand cuts.
That’s what virtually everyone agrees.
(I say “virtually” just to be safe, but I’ve never talked to or even heard of anyone who thinks that the current level of administrative requirements is actually optimal.)
This is why NIH-funded scientists regularly take to Twitter to say things like this:
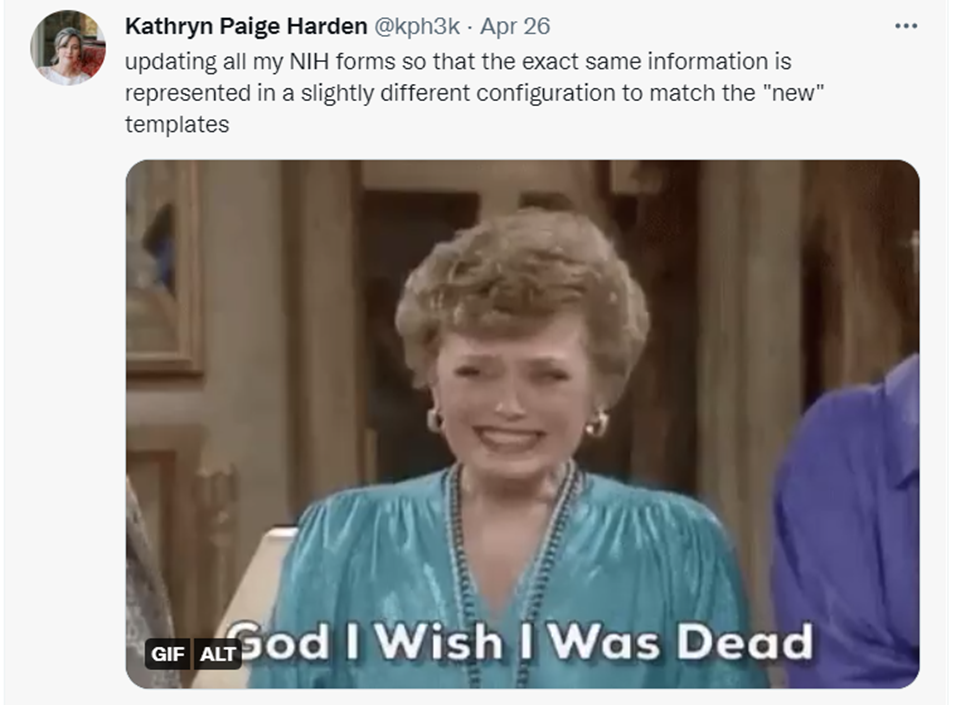
Or this:

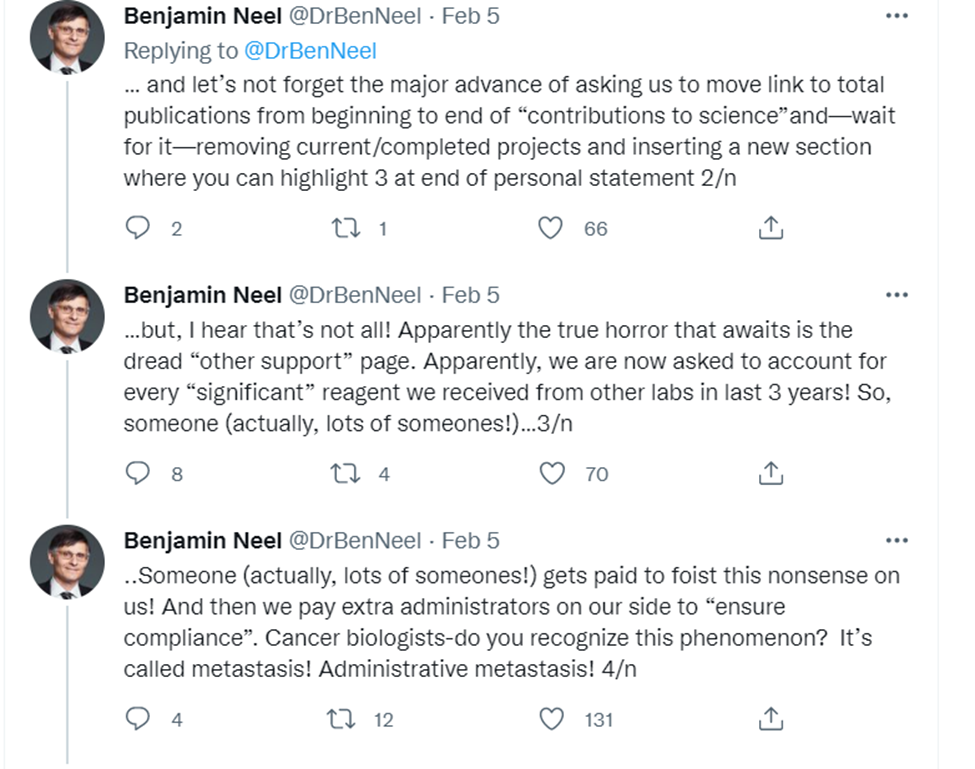
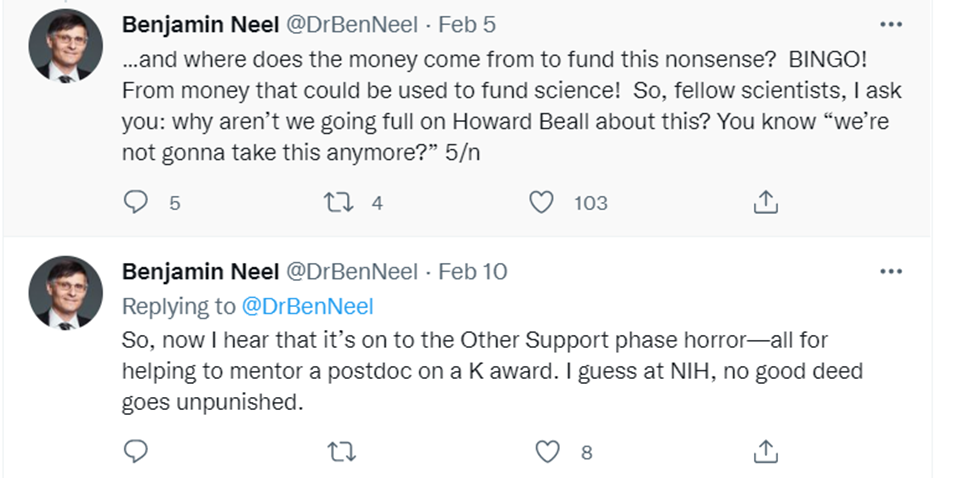
Or this:


Or this:
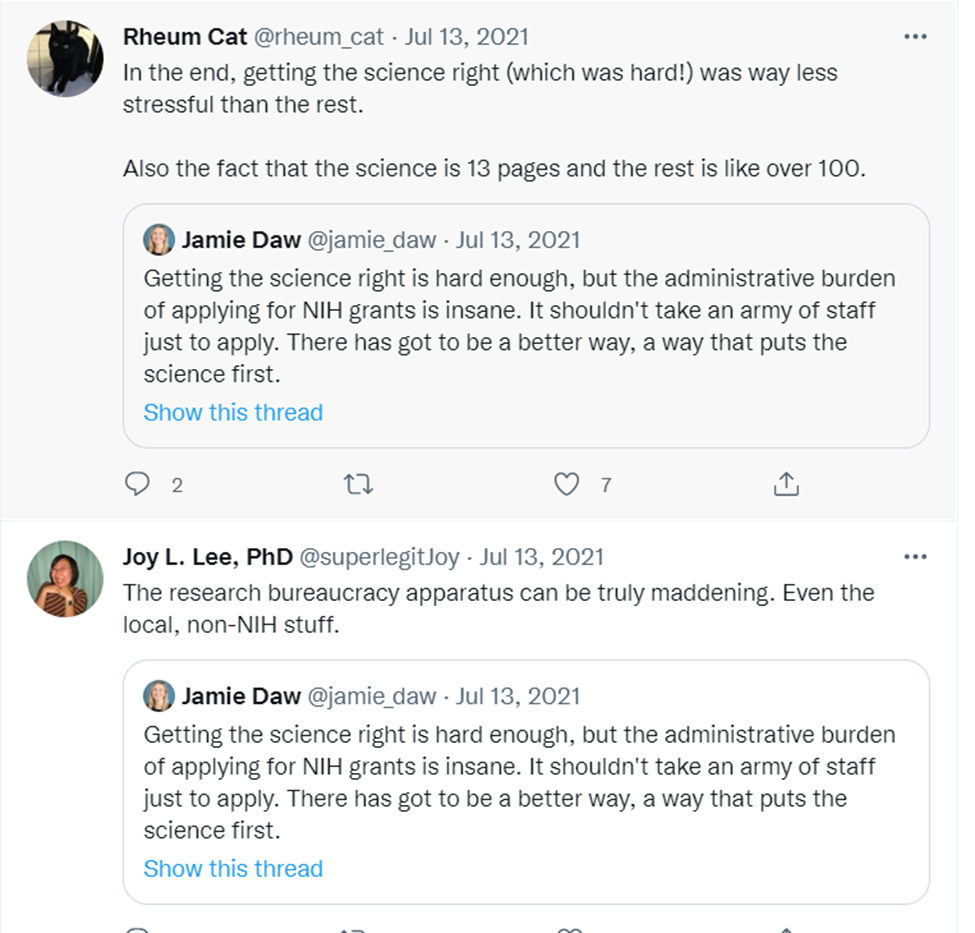
As Thomas Südhof of Stanford told me, “The biggest problem is that administrators . . . simply do not understand how much the bureaucratic work takes away from the science. They seem to think that just filling out another form or writing another report is nothing, but if you have to do hundreds of these, that is all you have time to do.”
When even Nobel laureates feel that way, imagine how early-career researchers feel.
We have to find a better way to handle federal science funding—fewer requirements, regulations, reporting.
We can’t keep making so many scientists tear out their hair over well-intentioned but near-infinite administrative requirements, as if that doesn’t distract from the actual science that they are supposed to be doing.
[…] the first installment of this series, I wrote about the burden of administrative compliance on federally-funded […]